More Information
Submitted: March 05, 2026 | Accepted: March 12, 2026 | Published: March 13, 2026
Citation: Moore RG, Smith E, Armbuster Y, Miller MG, Messerlian G, Blackman A. A Novel Marker for the Management of Patients Diagnosed with Endometrial Adenocarcinoma of the Uterus. Clin J Obstet Gynecol. 2026; 9(1): 001-007. Available from:
https://dx.doi.org/10.29328/journal.cjog.1001199
DOI: 10.29328/journal.cjog.1001199
Copyright license: © 2026 Moore RG, et al. This is an open access article distributed under the Creative Commons Attribution License, which permits unrestricted use, distribution, and reproduction in any medium, provided the original work is properly cited.
Keywords: Endometrial cancer; Serum HE4; Biomarker; Monitoring
A Novel Marker for the Management of Patients Diagnosed with Endometrial Adenocarcinoma of the Uterus
Richard G Moore1*, Elaine Smith1, Youngeun Armbuster1, M Craig Miller2, Geralyn Messerlian3 and Alexandra Blackman1
1Division of Gynecologic Oncology, Department of Obstetrics and Gynecology, Wilmot Cancer Institute, University of Rochester Medical Center, Rochester, New York, USA
2Fujirebio Diagnostics Inc, Malvern, PA, USA
3Department of Pathology, Women and Infants Hospital, Alpert Medical School of Brown University, Providence, RI, USA
*Corresponding author: Richard G Moore, MD, Division of Gynecologic Oncology, Department of Obstetrics and Gynecology, Wilmot Cancer Institute University of Rochester Medical Center, University of Rochester, Rochester, NY 14620, USA, Email: [email protected]
Background: Currently, there are no accurate clinical biomarkers for the management of patients diagnosed with endometrial cancer. The biomarker HE4 is often overexpressed and found to be elevated in the serum of patients with endometrial cancers.
Objective: The objective of this trial was to assess the utility of HE4 as a biomarker for monitoring patients with endometrial cancer.
Methods: This was an IRB-approved trial utilizing residual serial serum samples from patients diagnosed with endometrial cancer. Clinical status was determined by a combination of physician assessment, physical exam, serum CA125 levels, and imaging. Two separate analyses were performed to assess changes in HE4 serum levels. The first method used a change of 25% over that of the previous serum HE4 value, and the second analysis employed a velocity of change calculation of serum HE4 levels over time.
Results: A total of 92 patients providing 799 serum samples were identified for analysis. Using a ≥25.0% increase of serum HE4 levels as an indicator of disease recurrence or progression, HE4 had an accuracy of 81.9% and a negative predictive value of 91.4%. The velocity of change analysis an accuracy of 85.4% with a negative predictive value of 86.7%.
Conclusion: HE4 is highly accurate for detecting disease recurrence or progression and is a valuable clinical tool for monitoring patients with endometrial cancer.
Endometrial cancer is the fourth most common cancer diagnosed in women, resulting in approximately 13,860 deaths in 2025 in the US [1]. The incidence of endometrial cancer has been rising in the US and globally and will become one of the most common cancer diagnoses for women [2,3]. Endometrial cancer is associated with risk factors that raise levels of estrogen, such as obesity, unopposed menopausal estrogen therapy, polycystic ovarian syndrome, nulliparity, late menopause, and the use of Tamoxifen. While the 5-year survival rate is high at 81%, survival for patients with regional or distant metastatic disease dramatically decreases to 70% and 19%, respectively [1].
Endometrial cancer has historically been characterized into two groups. Type I is the most common and is usually associated with excess estrogen and made up of the endometrioid histologic subtype. Ten to fifteen percent of endometrial carcinomas are classified as Type II and include serous, clear cell, carcinosarcoma, and undifferentiated carcinomas. These tumors tend to arise in older patients, do not express estrogen or progesterone receptors, and have a poor prognosis. Approximately 13% of endometrial carcinoma patients will experience recurrence of their disease, usually within three years; however, only 3% of patients with low-risk cancers will recur [4]. Patients with a symptomatic recurrence tend to have improved outcomes compared with patients found with recurrences without symptoms [5].
Methods for the detection of endometrial cancer in symptomatic patients with postmenopausal vaginal bleeding or vaginal discharge include transvaginal ultrasound, endometrial cytology, endometrial biopsy, or surgical sampling through endometrial curettage with or without hysteroscopy [6]. There are no effective non-invasive methods of screening for endometrial cancer available for asymptomatic patients. Similarly, no consistent guidelines are available to guide the post-surgical follow-up of patients diagnosed with endometrial cancer. Currently, only physical examination is recommended for the follow-up of patients after surgery and or adjuvant treatment, with the value of imaging, blood testing, and vaginal smears being questioned [7-9].
Several serum biomarkers have been investigated for diagnosis and prognosis in patients with endometrial carcinoma, including CA125. While pre-treatment CA1be predictive of stage, depth of myometrial invasion, grade, and other indicators of outcome [10], the sensitivity of CA125 for the detection of endometrial cancer is low, particularly in early-stage patients and in asymptomatic patients with recurrent disease [10-12]. Other biomarkers have been elevated in patients with endometrial cancer, including trefoil factor 3, calprotectin, and amyloid A, but these have not been evaluated serially in patients who have recurrent or progressive disease [13-15].
We previously evaluated human epididymal protein 4 (HE4), a whey acidic four disulfide core protein, in the serum of patients with endometrial cancer and were the first to show that HE4 is elevated across all stages of endometrial cancer [16], and that preoperative serum HE4 levels can predict depth of myometrial invasion [17]. Several subsequent studies have confirmed that serum levels of HE4 are elevated in patients with endometrial cancer compared to healthy controls [18-20] and that preoperative serum levels of HE4 are a prognostic indicator for overall survival and progression-free survival [18,20,21]. We were also the first to demonstrate that serum HE4 levels change over time in patients with endometrial cancer with disease progression and regression [22]. In this study, we evaluate longitudinal changes in serum HE4 concentrations post-surgically for the detection of recurrence or progression of endometrial cancer in patients undergoing treatment for disseminated disease.
The data collected for this study were reviewed and approved by the Wilmot Cancer Institute of the University of Rochester IRB (RSRBB00063471), and the serum sample collection for this study was reviewed and approved by the Women and Infants Hospital of Rhode Island (WIHRI) IRB (Project No. 09-0334). Clinical residual serum samples from patients diagnosed with endometrial cancer that had been on treatment or undergoing surveillance from J1997 to October 2010 had been stored at -80 °C and were available through the WIHRI Division of Medical Screening and Special Testing. Longitudinal serum samples were obtained from patients undergoing routine laboratory testing and evaluated retrospectively. Blood was drawn at various times before and after surgery, during chemotherapy treatment, and in follow-up monitoring in patients with histologically confirmed endometrial carcinoma. Blood samples were centrifuge and the serum was then aliquoted and stored at -80 °C within four hours of collection. Patients included in the study were actively being treated with chemotherapy for metastatic disease or being monitored for recurrence after completing chemotherapy. All patients in this study were being followed clinically with serial CA125 as part of their routine clinical evaluation and monitoring. Longitudinal sets of specimens included all serial draws during the following time : i) post-surgery to the time of first recurrence, ii) post-surgery over time with no evidence of recurrence, iii) during chemotherapeutic treatment for recurrent or persistent disease, or iv) post-chemotherapeutic treatment during the monitoring phase. Clinical status was determined at the time of each blood draw by querying the medical rphysician's assessment using physical examination, serum CA125 concentrations, and imaging. Outcomes measured included clinical assessment for recurrence, progression, regression, or stable disease.
HE4 was measured by trained technicians blinded to the clinical status of the subjects using the Fujirebio HE4 kit (Fujirebio Diagnostics Inc., Malvern, PA). Specimens were run in duplicate, and control specimens were within pre-specified ranges for each run of patient sera according to the manufacturer’s instructions.
Statistical analysis
All pinto the study had to have a minimum of 3 serial serum samples available for evaluation. Patients undergoing chemotherapy treatment were clinically evaluated to either have progression or regression of disease. Patients undergoing monitoring were evaluated and categorized as having either progressive disease (disease progression or recurrence) or non-progressive disease (disease regression, stable disease, or no evidence of disease). This allowed for a binary analysis of the serum biomarkers as either progressive disease or non-progressive disease. Two separate statistical analyses were performed to evaluate changes in serum HE4 concentrations over time. The first analysis used a single point in time increase of serum HE4 levels of ≥ 25.0% as compared to the previous serum HE4 value, with an increase of ≥ 25.0% being considered an indication of disease progression. The second analysis employed the velocity of change for serum HE4 levels over time using a minimum of 3 serial samples to determine progression versus non-progression. The velocity of change analysis of serial samples employed thresholds of ≥10.0 pM per month for serum HE4 levels.
Serum HE4 biomarker levels were compared to the clinical truths as determined by clinical assessment by the attending physician, which included any combination of physical exam, CA125 biomarker levels, and imaging. For statistical purposes, two clinical outcome groups were created: no progression (patients with no evidence of disease, stable disease, or regressive disease) and progression (patients with new recurrent disease or progressive disease).
An additional analysis was performed to determine the lead time with which HE4 and CA125, alone or in combination, detected recurrence or progression of disease. For any set of longitudinal biomarker results for a given patient, a rise of > 25% above the nadir reached at or following the start of treatment was considered an indication of recurrence or progression.
A total of 799 serum samples were obtained from 92 different patients. These specimens comprised 123 longitudinal sets, each of which had at least 4 serial samples for analysis (range = 4 to 12 draws, median = 7). Of the 123 sets of serial specimens, 85 (69.1%) consisted of specimens collected while patients were on chemotherapy treatments, and 38 (30.9%) consisted of specimens collected while patients were undergoing surveillance. The demographics of the study cohort are displayed in Table 1. The median age was 64 years (range 38 - 92), and 31.5% of patients had endometrioid histology, 42.4% of patients had serous histology, and 26.1% were clear cell, mixed, or other histologic subtypes. There were 31 patients (33.7%) with surgical stage I disease, 12 (13.0%) with stage II, 33 (35.9%) with stage III, and 16 (17.4%) with stage IV disease. The breakdown of tumor grade showed 10 patients (10.9%) had grade 1 tumors, 15 (16.3%) grade 2, and 67 (72.8%) grade 3 tumors.
| Table 1: Patient demographics* | ||
| Age at Diagnosis | Years | |
| Average (Median) age | 65 (64) | |
| Range years old | 38 - 92 | |
| Histology | N | (%) |
| Endometrioid | 29 | (31.5) |
| Serous / Papillary Serous | 39 | (42.4) |
| Carcinosarcoma | 5 | (5.4) |
| Clear Cell | 4 | (4.3) |
| Clear Cell & Mixed | 1 | (1.1) |
| Endometrioid & Clear Cell | 4 | (4.3) |
| Endometrioid & PNET component | 1 | (1.1) |
| Serous & Clear Cell | 3 | (3.3) |
| Serous & Endometrioid | 6 | (6.5) |
| Tumor Stage | N | (%) |
| Stage I | 31 | (33.7) |
| Stage II | 12 | (13.0) |
| Stage III | 33 | (35.9) |
| Stage IV | 16 | (17.4) |
| Tumor Grade | N | (%) |
| Grade 1 | 10 | (10.9) |
| Grade 2 | 15 | (16.3) |
| Grade 3 | 67 | (72.8) |
| Treatment Status | Patients (N) | |
| On Treatment | 85 | (69.1) |
| Follow-up | 38 | (30.9) |
| Total | 123 | (100) |
| *Includes 92 patients and 123 longitudinal data sets. | ||
We evaluated the accuracy of serial measurements of serum HE4 levels to predict progressive versus non-disease in the 123 specimen sets using a threshold of a ≥25% increase from sample to sample (Table 2) or a velocity of change of ≥10.0 pM per month as described above (Table 3).
| Table 2: Performance characteristics of HE4 in the monitoring of patients with endometrial cancer using a threshold of ≥25% increase from sample to sample based on histologic subtypes. | |||||
| Histology (# of Treatment Sets) |
Accuracy (CI 95%) |
NPV (CI 95%) |
PPV (CI 95%) |
Specificity (CI 95%) |
Sensitivity (CI 95%) |
| All (N = 123) | 81.9% (78.7 - 84.8) |
91.4% (88.6 - 93.6) |
30.6% (21.7 - 40.7) |
87.7% (84.7 - 90.3) |
39.5% (28.4 - 51.4) |
| Endometrioid (N = 39) | 83.6% (77.6 - 88.5) |
94.0% (89.3 - 97.1) |
21.4% (8.3 - 41.0) |
87.7% (82.0 - 92.1) |
37.5% (15.2 - 64.6) |
| Serous (N = 71) | 81.3% (76.9 - 85.1) |
89.2% (85.1 - 92.4) |
39.7% (27.0 - 53.4) |
88.6% (84.5 - 91.9) |
41.1% (28.1 - 55.0) |
| All Others (N = 13) | 80.6% (69.5 - 88.9) |
95.0% (86.1 - 99.0) |
8.3% (0.2 - 38.5) |
83.8% (72.9 - 91.6) |
25.0% (0.6 - 80.6) |
| Table 3: Performance characteristics of HE4 in the monitoring of patients with endometrial cancer using a velocity of change of ≥10.0 pM per month based on histologic subtypes. | |||||
| Histology (# of Treatment Sets) |
Accuracy (CI 95%) |
NPV (CI 95%) |
PPV (CI 95%) |
Specificity (CI 95%) |
Sensitivity (CI 95%) |
| All (N = 123) | 85.4% (77.9 - 91.1) |
86.7% (78.6 - 92.5) |
77.8% (52.4 - 93.6) |
95.8% (89.6 - 98.8) |
50.0% (30.6 - 69.4) |
| Endometrioid (N = 39) | 92.3% (79.1 - 98.4) |
94.1% (80.3 - 99.3) |
80.0% (28.4 - 99.5) |
97.0% (84.2 - 99.9) |
66.7% (22.3 - 95.7) |
| Serous (N = 71) | 80.3% (69.1 - 88.8) |
81.4% (69.1 - 90.3) |
75.0% (42.8 - 94.5) |
94.1% (83.8 - 98.8) |
45.0% (23.1 - 68.5) |
| All Others (N = 13) | 92.3% (64.0 - 99.8) |
91.7% (61.5 - 99.8) |
100.0% (2.5 - 100.0) |
100.0% (71.5 - 100.0) |
50.0% (1.3 - 98.7) |
Examination of all histologic types of endometrial cancer using the single point in time analysis with a serum HE4 change of a ≥ 25.0% increase provided an accuracy of 81.9% (CI 95%: 78.7% - 84.8%), a negative predictive value of 91.4% (CI 95%: 88.6% - 93.6%), a specificity of 87.7% (CI: 84.7% - 90.3%), and a sensitivity of 39.5% (CI 95%: 28.4% - 51.4%).
A velocity of change in serum HE4 levels of ≥10.0 pM per month was found to optimize the statistical performance of changes in serum HE4 levels when analysed over time. When examining all histologic subtypes together using a threshold of ≥10.0 pM per month for the velocity of change in serum HE4 levels, the changes in HE4 were able to predict progressive versus non-progressive disease with an accuracy of 85.4% (CI 95%: 77.9% - 91.1%), a negative predictive value of 86.7% (CI 95%: 78.6% - 92.5%), a specificity of 95.8% (CI 95%: 89.6% - 98.8%), and a sensitivity of 50.0% (CI 95%: 30.6% - 69.4%).
We then evaluated histologic subtypes separated into the categories of endometrioid, serous (serous and mixed serous), and others (clear cell, carcinosarcoma) as detailed in Tables 2 and 3. Examination of a point to point change in serum HE4 levels of ≥ 25.0% to predict progressive versus non-progressive disease in the different histologic subtypes showed the following: For endometrioid histology alone, an accuracy of 83.6% (CI 95%: 77.6% - 88.5%), a negative predictive value of 94.0% (CI 95%: 89.3% - 97.1%), a specificity of 87.7% (CI 95%: 82.0% - 92.1%), and a sensitivity of 37.5% (CI 95%: 15.2% - 64.6%) were obtained. For serous histology, an accuracy of 81.3% (CI 95%: 76.9% - 85.1%), a negative predictive value of 89.2% (CI 95%: 85.1% - 92.4%), a specificity of 88.6% (CI 95%: 84.5% - 91.9%), and a sensitivity of 41.1% (CI 95%: 28.1% - 55.0%) were obtained. For all other histological subtypes, an accuracy of 80.6% (CI 95%: 69.5% - 88.9%), a negative predictive value of 95.0% (CI 95%: 86.1% - 99.0%), a specificity of 83.8% (CI 95%: 72.9% - 91.6%) and a sensitivity of 25.0% (CI 95%: 0.6% - 80.6%) were determined.
Examination of the velocity of change in serum HE4 levels of ≥10.0 pM per month to predict progressive versus non-progressive disease in the different histologic subtypes revealed the following: For endometrioid histology alone, an accuracy of 92.3% (CI 95%: 79.1% - 98.4%), a negative predictive value of 94.1% (CI 95%: 80.3% - 99.3%), a specificity of 97.0% (CI 95%: 84.2% - 99.9%), and a sensitivity of 66.7% (CI 95%: 22.3% - 95.7%) were obtained. For serous histology, an accuracy of 80.3% (CI 95%: 69.1% - 88.8%), a negative predictive value of 81.4% (CI 95%: 69.1% - 90.3%), a specificity of 94.1% (CI 95%: 83.8% - 98.8%), and a sensitivity of 45.0% (CI 95%: 23.1% - 68.5%) were obtained. For all other histological subtypes, an accuracy of 92.3% (CI 95%: 64.0% - 99.8%), a negative predictive value of 91.7% (CI 95%: 61.5% - 99.8%), a specificity of 100.0% (CI 95%: 2.5% - 100.0%) and a sensitivity of 50.0% (CI 95%: 1.3% - 98.7%) were determined.
A comparative analysis between the performance characteristics of the velocities of change in HE4 and CA125 to predict progressive versus non-progressive disease was performed using a set sensitivity of 50.0%. This resulted in a velocity of change threshold of ≥1.0 U/mL per month for CA125 and ≥10.0 pM per month for HE4. Despite a bias in favor of CA125, as this biomarker was part of the clinical assessment evaluation, a useful comparison can be made. At a set sensitivity 50.0% (CI 95%: 30.6% - 69.4%), HE4 had an accuracy of 85.4% (CI 95%: 77.9% - 91.1%), a negative predictive value of 86.7% (95% CI: 78.6% to 92.5%), and a specificity of 95.8% (95% CI: 89.6% to 98.8%), compared to CA125 which had an accuracy of 84.6% (CI 95%: 76.9% - 90.4%), a negative predictive value of 86.5% (95% CI: 78.4% to 92.4%), and a specificity of 94.7% (95% CI: 88.1% to 98.3%).
The comparison between HE4 and CA125 velocity of changes at a set sensitivity of 50.0% showed no significant differences between the ability of CA125 or HE4 to predict progressive versus non-progressive disease when including all histological subtypes. HE4 performed equally as well despite CA125 being used in the determination of the clinical status of the patients in this cohort. An analysis using an either/or combination of the HE4 and CA125 velocity of changes at the same thresholds as above showed an accuracy of 87.8% (CI 95%: 80.7% - 93.0%), a negative predictive value of 90.8% (CI 95%: 83.3% - 95.7%), a specificity of 93.7% (CI 95%: 86.8% - 97.6%), and a sensitivity of 67.9% (CI 95%: 47.6% - 84.1%). The dual marker combination showed a 17.9% improvement in sensitivity while maintaining a high overall agreement, negative predictive value, and specificity.
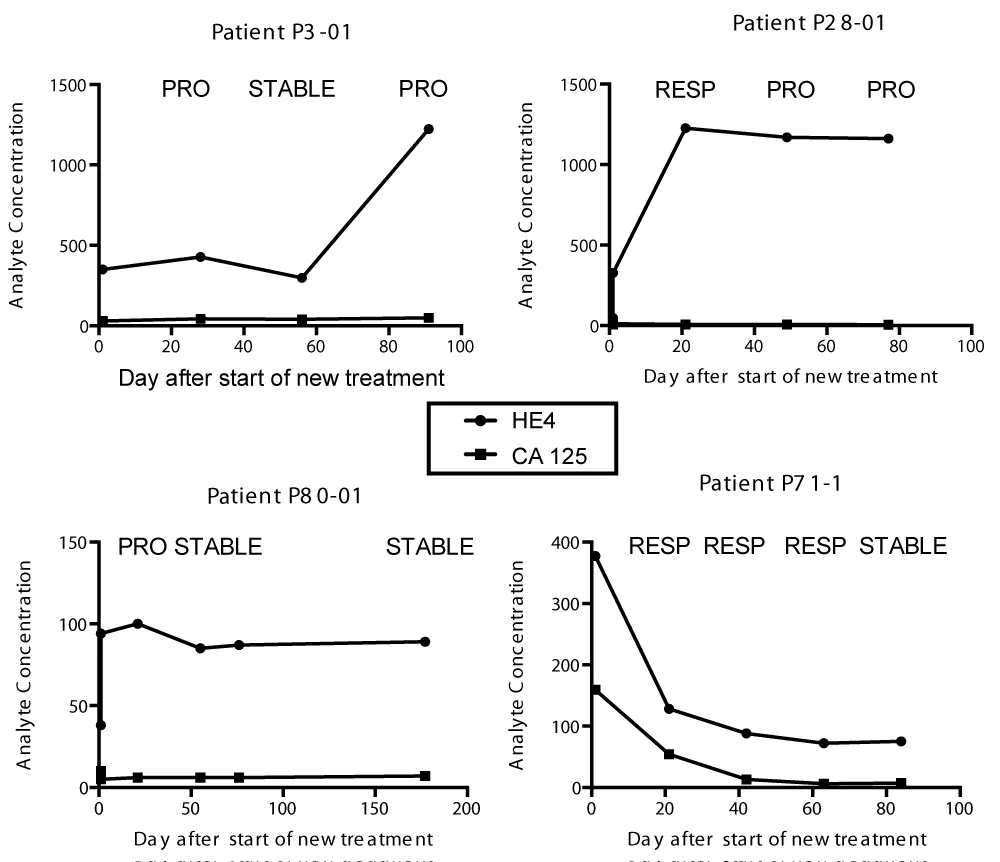
Changes over time included three potential patterns: for some patients, the serial values rose prior to or at the time progression was noted clinicallpatients, serial values remained unchanged during periods of stable disease; and in some patients, serial values decreased over time with clinical response to therapy. These types of changes are illustrated graphically in Figure 1.
Figure 1: Illustration of patterns of HE4 and CA125 serum biomarker changes over time.
Graphical presentation of serial data showed that for some serial sets, HE4 showed increasing serum levels over time prior to progression, whereas CA125 values did not rise, as with data sets P3-01 and P28-01 in Figure 1. For other serial sets, neither biomarker provided useful longitudinal changes, as demonstrated in P80-01, and for some serial sets, both biomarkers provided interpretable longitudinal changes, as demonstrated in P71-1. Therefore, we evaluated longitudinal changes in both biomarkers over time to define lead time for the prediction of progression. There were 30 serial sets of specimens with a baseline blood draw, a minimum of two additional blood draws following the start or at the time that progression was noted clinically. Using a 25% increase over the nadir concentration for each biomarker, HE4 detected progression prior to the time of clinical progression in 40.0% (12/30) of the serial sets, whereas CA125 detected progression prior to the time of clinical progression in 26.7% (8/30) of the serial sets (Table 4). The mean lead times for detection of progression were 59 days for HE4 (range 28 - 371 days) and 34 days for CA 125 (range 61 - 203 days). For both biomarkers combined, the lead time for early detection of progression was 53 days in 50.0% (15/30) of the serial sets.
| Table 4. Lead time for detection of progression or recurrence of endometrial cancer using longitudinal HE4 and CA125 compared with clinical and or CT imaging detection of recurrence/progression. |
|||
| Patient ID | HE4 Lead Time (Days)1 | CA 125 Lead Time (Days) | Combined Lead Time (Days) |
| P2-02 | - | 0 | 0 |
| P3-01 | 0 | - | 0 |
| P3-02 | 28 | 0 | 28 |
| P3-03 | 0 | 0 | 0 |
| P8-01 | - | 0 | 0 |
| P8-02 | 37 | - | 37 |
| P8-03 | - | 0 | 0 |
| P11-01 | 0 | 0 | 0 |
| P16-01 | 83 | 0 | 83 |
| P19-01 | 123 | - | 123 |
| P21-01 | 0 | 0 | 0 |
| P22-01 | 42 | 71 | 71 |
| P24-02 | 203 | 203 | 203 |
| P25-01 | - | 0 | 0 |
| P28-01 | 28 | - | 28 |
| P36-01 | - | 0 | 0 |
| P42-01 | - | 0 | 0 |
| P43-01 | 371 | 0 | 371 |
| P45-02 | 78 | 78 | 78 |
| P48-01 | - | 91 | 91 |
| P56-01 | - | 0 | 0 |
| P57-01 | 63 | 0 | 63 |
| P60-01 | 0 | 0 | 0 |
| P62-01 | - | 108 | 108 |
| P65-02 | - | 161 | 161 |
| P69-01 | 0 | - | 0 |
| P71-02 | 0 | 0 | 0 |
| P74-01 | 61 | 61 | 61 |
| P75-02 | 70 | 70 | 70 |
| P77-01 | 0 | 0 | 0 |
| N With Lead Time | 12 / 30 (40.0%) | 8 / 30 (26.7%) | 15 / 30 (50.0%) |
| Mean Lead Time | 59 Days | 34 Days | 53 Days |
| Range (Days) | 28 - 371 | 61 - 203 | 28 - 371 |
| Biomarkers were considered indicative of recurrence/progression when values increased > 25% above nadir concentrations following the start of treatment. Blank entries (-) indicate that the biomarker did not detect recurrence/progression in that patient, whereas a 0 (zero) entry indicates detection of recurrence/progression with no lead time compared with detection using traditional methods (clinical impression or CT imaging). |
|||
Biomarkers were considered indicative of recurrence/progression when values increased > 25% above nadir concentrations following the start of treatment. Blank entries (-) indicate that the biomarker did not detect recurrence/progression in that patient, whereas a 0 (zero) entry indicates detection of recurrence/progression with no lead time compared with detection using traditional methods (clinical impression or CT imaging).
Patients diagnosed with high-grade endometrioid or serous endometrial carcinomas will experience disease recurrence and progression, and long-term survival for these patients is low. Current modalities of surveillance, including imaging, serum CA125 measurement, and physical examination, may miss asymptomatic recurrences, and the long-term survival of patients whose recurrence is detected while asymptomatic is greater than for patients who experience symptomatic recurrences [7]. For these reasons, better tools are needed to monitor patients with high-risk endometrial cancers for recurrence and progression.
Be an effective biomarker for monitoring patients with ovarian cancer and is FDA cleared for monitoring patients with epithelial ovarian cancer [12]. HE4 has also been shown to provide a high sensitivity for the detection of endometrial cancer and as a prognostic indicator [11,16]. Preoperative serum HE4 concentrations have been shown to correlate with depth of myometrial invasion and are a prognostic indicator for overall survival and progression-free survival [17-20]. This may be explained by recent data showing that silencing of the HE4 gene inhibits cell proliferation, migration, and invasion in endometrial cancer cell lines [23]. Conversely, overexpression of HE4 enhanced cell proliferation and invasion in vitro, and promoted tumor growth in a mouse xenograft model of ovarian cancer [23].
In the current study, HE4 demonstrated a high accuracy, specificity, and negative predictive value for the detection of recurrence and progression in patients undergoing surveillance and or treatment for endometrial cancer. We evaluated two methods for the detection of recurrence or progression using changes in serum HE4 levels over time, with a ≥ 25% increase in biomarker values from point to point, or a velocity of change of ≥ 10.0 pg/mL/month across all samples. Serum HE4 measurements showed increasing concentrations in patients with progressive disease, decreasing concentrations in patients responding to systemic therapy, and stable concentrations in patients with stable disease. Although our longitudinal analyses were confined to a binary approach using progression versus no progression, trends of HE4 over time may be useful to follow treatment response as well as for the detection of disease recurrence or progression. While the accuracy, defined as the agreement of the test with the patients’ clinical status, was high, equally important was the high negative predictive value of 91.4%. This suggests that HE4 may be a useful biomarker enabling the delay of imaging until the biomarker values show a rising trend, thus saving cost and discomfort to patients.
Of the 92 patients that provided serum samples for this study, 31.5% had endometrioid endometrial carcinoma. While the proportion of endometrioid cancers is typically higher in newly diagnosed patients, the proportion with serous carcinoma would be expected to be higher in a cohort of patients who experience recurrence. In this study, HE4 was elevated in patients with both endometrioid endometrial cancer and in patients with serous endometrial cancer. However, HE4 sensitivities in patients with these histological subtypes of endometrial carcinoma have not been well studied. This study demonstrates HE4 has a higher accuracy by 12% in patients with endometrioid histology versus serous histology (92.3% vs. 80.3%) and is an accurate marker for endometrioid histologic types of endometrial cancers. One prior study found that HE4 is a better indicator of recurrence than CA125 for endometrioid histology (AUC 0.87 vs. 0.67), but this superiority was not seen in other histologies [24] be elevated across all stages and grades of endometrial cancer in some studies [16,20] but was stage and grade-dependent in others [18,25]. In our cohort, we also saw that HE4 statistical performance was not inferior to CA125 across all histological subtypes, despite a bias towards CA125 in this study.
Other recent studies have found elevated HE4 to be an independent risk factor for lymph node invasion, deep myometrial invasion, and primary tumor diameter [24,26]. Overall, the relationship between HE4 serum levels and stage and grade requires further investigation.
The serum biomarker CA125 showed similar performance to HE4 in monitoring for recurrence and progression of endometrial cancer in this study, and both biomarkers showed increasing, decreasing, and stable trends over time in different patients (Figure 1). We therefore evaluated both biomarkers for their ability to detect progression or recurrence with a lead time. We found recurrence/progression before detection by standard practice in approximately 50% of patients (Table 4). HE4 detected recurrence/progression with an average lead time of 59 days in 40% of evaluable patients, and CA125 was positive with an average lead time of 34 days in 27% of patients. However, the biomarkers detected recurrence/progression in a different set of patients with different lead times. Thus, if either of the two biomarkers showed a rise of ≥ 25% or more from the nadir value, 50% of patients could be dto conventional methods, with an average lead time of 53 days. This suggests that the use of both CA125 and HE4 in combination is more effective for monitoring for recurrence and progression in patients with endometrial cancer than the use of either biomarker alone. This is consistent with the use of HE4 and CA125 combined in the risk of ovarian malignancy (ROMA) algorithm [27].
The strength of this study is the large number of serial serum samples from patients with endometrial cancer that were available to assess the change in disease status over time. A weakness of this trial is that the clinical status of patients was determined by a number of variable factors that could not be standardized for all patients, given the retrospective nature of the trial.
Our results suggest that the serum biomarker HE4 can be an effective tool to monitor patients with endometrial cancer for recurrence of cancer following primary surgery, and for the detection of progression while on treatment for recurrent or progressive disease. HE4 is also useful to monitor response to therapy and to confirm stable and responding disease, particularly in the histological subtype of endometrioid adenocarcinoma. Given the high negative predictive value, serial concentrations of HE4 that do not increase more than 25% from the previous value may be used to safely delay more expensive and invasive imaging procedures. The utilization of measuring serum HE4 levels provides a useful tool for monitoring patients with endometrial cancer.
Conflict of interest: RGM receives research funding from Fujirebio Diagnostics Inc. MCM is an employee of Fujirebio Diagnostics Inc.
Funding: This Study was funded by Fujirebio Diagnostics Inc.
Ethical approval: IRB review. This study was exempt from federal regulation under the following category (45 CFR 46.101) as the study used pre-existing deidentified data. Therefore, there are no consent forms for this study as the data used was de-identified and pre-existing.
- Siegel RL, Kratzer TB, Giaquinto AN, Sung H, Jemal A. Cancer statistics, 2025. CA Cancer J Clin. 2025;75(1):10-45. Available from: https://doi.org/10.3322/caac.21871
- Fader AN, Arriba LN, Frasure HE, von Gruenigen VE. Endometrial cancer and obesity: epidemiology, biomarkers, prevention and survivorship. Gynecol Oncol. 2009;114(1):121-7. Available from: https://doi.org/10.1016/j.ygyno.2009.03.039
- Wartko P, Sherman ME, Yang HP, Felix AS, Brinton LA, Trabert B. Recent changes in endometrial cancer trends among menopausal-age U.S. women. Cancer Epidemiol. 2013;37(4):374-7. Available from: https://doi.org/10.1016/j.canep.2013.03.008
- Fung-Kee-Fung M, Dodge J, Elit L, Lukka H, Chambers A, Oliver T. Follow-up after primary therapy for endometrial cancer: a systematic review. Gynecol Oncol. 2006;101(3):520-9. Available from: https://doi.org/10.1016/j.ygyno.2006.02.011
- Gadducci A, Cosio S, Genazzani AR. Tissue and serum biomarkers as prognostic variables in endometrioid-type endometrial cancer. Crit Rev Oncol Hematol. 2011;80(2):181-92. Available from: https://doi.org/10.1016/j.critrevonc.2010.11.005
- Clark TJ, Barton PM, Coomarasamy A, Gupta JK, Khan KS. Investigating postmenopausal bleeding for endometrial cancer: cost-effectiveness of initial diagnostic strategies. BJOG. 2006;113(5):502-10. Available from: https://doi.org/10.1111/j.1471-0528.2006.00914.x
- Carrara L, Gadducci A, Landoni F, Maggino T, Scambia G, Galletto L, et al. Could different follow-up modalities play a role in the diagnosis of asymptomatic endometrial cancer relapses?: an Italian multicentric retrospective analysis. Int J Gynecol Cancer. 2012;22(6):1013-9. Available from: https://doi.org/10.1097/igc.0b013e31825ad3ee
- Querleu D, Planchamp F, Narducci F, Morice P, Joly F, Genestie C, et al. Clinical practice guidelines for the management of patients with endometrial cancer in France: recommendations of the Institut National du Cancer and the Société Française d'Oncologie Gynécologique. Int J Gynecol Cancer. 2011;21(5):945-50. Available from: https://doi.org/10.1097/igc.0b013e31821bd473
- Sartori E, Pasinetti B, Chiudinelli F, Gadducci A, Landoni F, Maggino T, et al. Surveillance procedures for patients treated for endometrial cancer: a review of the literature. Int J Gynecol Cancer. 2010;20(6):985-92. Available from: https://doi.org/10.1111/igc.0b013e3181e2abcc
- Chao A, Tang YH, Lai CH, Chang CJ, Chang SC, Wu TI, et al. Potential of an age-stratified CA125 cut-off value to improve the prognostic classification of patients with endometrial cancer. Gynecol Oncol. 2013;129(3):500-4. Available from: https://doi.org/10.1016/j.ygyno.2013.02.032
- Angioli R, Plotti F, Capriglione S, Montera R, Damiani P, Ricciardi R, et al. The role of novel biomarker HE4 in endometrial cancer: a case-control prospective study. Tumour Biol. 2013;34(1):571-6. Available from: https://doi.org/10.1007/s13277-012-0583-0
- Li J, Dowdy S, Tipton T, Podratz K, Lu WG, Xie X, et al. HE4 as a biomarker for ovarian and endometrial cancer management. Expert Rev Mol Diagn. 2009;9(6):555-66. Available from: https://doi.org/10.1586/erm.09.39
- Bignotti E, Ravaggi A, Tassi RA, Calza S, Rossi E, Falchetti M, et al. Trefoil factor 3: a novel serum marker identified by gene expression profiling in high-grade endometrial carcinomas. Br J Cancer. 2008;99(5):768-73. Available from: https://doi.org/10.1038/sj.bjc.6604546
- Cocco E, Bellone S, El-Sahwi K, Cargnelutti M, Buza N, Tavassoli FA, et al. Serum amyloid A: a novel biomarker for endometrial cancer. Cancer. 2010;116(4):843-51. Available from: https://doi.org/10.1002/cncr.24838
- Ni Bhriain H, Trovik J, Wik E, Stefansson IM, Akslen LA, Salvesen HB, et al. Plasma calprotectin concentrations in women with endometrial carcinoma. Gynecol Oncol. 2009;114(3):491-5. Available from: https://doi.org/10.1016/j.ygyno.2009.06.008
- Moore RG, Brown AK, Miller MC, Badgwell D, Lu Z, Allard WJ, et al. Utility of a novel serum tumor biomarker HE4 in patients with endometrioid adenocarcinoma of the uterus. Gynecol Oncol. 2008;110(2):196-201. Available from: https://doi.org/10.1016/j.ygyno.2008.04.002
- Moore RG, Miller CM, Brown AK, Robison K, Steinhoff M, Lambert-Messerlian G. Utility of tumor marker HE4 to predict depth of myometrial invasion in endometrioid adenocarcinoma of the uterus. Int J Gynecol Cancer. 2011;21(7):1185-90. Available from: https://doi.org/10.1097/igc.0b013e3182229ad8
- Bignotti E, Ragnoli M, Zanotti L, Calza S, Falchetti M, Lonardi S, et al. Diagnostic and prognostic impact of serum HE4 detection in endometrial carcinoma patients. Br J Cancer. 2011. Available from: https://doi.org/10.1038/bjc.2011.109
- Kalogera E, Scholler N, Powless C, Weaver A, Drapkin R, Li J, et al. Correlation of serum HE4 with tumor size and myometrial invasion in endometrial cancer. Gynecol Oncol. 2012;124(2):270-5. Available from: https://doi.org/10.1016/j.ygyno.2011.10.025
- Zanotti L, Bignotti E, Calza S, Bandiera E, Ruggeri G, Galli C, et al. Human epididymis protein 4 as a serum marker for diagnosis of endometrial carcinoma and prediction of clinical outcome. Clin Chem Lab Med. 2012;50(12):2189-98. Available from: https://doi.org/10.1515/cclm-2011-0757
- Mutz-Dehbalaie I, Egle D, Fessler S, Hubalek M, Fiegl H, Marth C, et al. HE4 is an independent prognostic marker in endometrial cancer patients. Gynecol Oncol. 2012;126(2):186-91. Available from: https://doi.org/10.1016/j.ygyno.2012.04.022
- Moore R, Horan T, Singh R, Kawar N, Eklund E, Lambert-Messerlian G, McCourt C, Hawkins D, Granai CO. A novel marker for the management of women diagnosed with endometrial adenocarcinoma of the uterus. Gynecol Oncol. 2013;130:e90. Available from: https://doi.org/10.1016/j.ygyno.2013.04.269
- Zou SL, Chang XH, Ye X, Cheng HY, Cheng YX, Tang ZJ, et al. Effect of human epididymis protein 4 gene silencing on the malignant phenotype in ovarian cancer. Chin Med J (Engl). 2011;124(19):3133-40. Available from: https://pubmed.ncbi.nlm.nih.gov/22040568/
- Stiekema A, Lok C, Korse CM, van Driel WJ, van der Noort V, Kenter GG, et al. Serum HE4 is correlated with prognostic factors and survival in patients with endometrial cancer. Virchows Arch. 2017;470(6):655-64. Available from: https://doi.org/10.1007/s00428-017-2115-1
- Omer B, Genc S, Takmaz O, Dirican A, Kusku-Kiraz Z, Berkman S, et al. The diagnostic role of human epididymis protein 4 and serum amyloid-A in early-stage endometrial cancer patients. Tumour Biol. 2013;34(5):2645-50. Available from: https://doi.org/10.1007/s13277-013-0814-z
- Wang Y, Han C, Teng F, Bai Z, Tian W, Xue F. Predictive value of serum HE4 and CA125 concentrations for lymphatic metastasis of endometrial cancer. Int J Gynaecol Obstet. 2017;136(1):58-63. Available from: https://doi.org/10.1002/ijgo.12010
- Moore RG, Miller MC, Disilvestro P, Landrum LM, Gajewski W, Ball JJ, et al. Evaluation of the diagnostic accuracy of the risk of ovarian malignancy algorithm in women with a pelvic mass. Obstet Gynecol. 2011;118(2 Pt 1):280-8. Available from: https://doi.org/10.1097/aog.0b013e318224fce2